 Water dissociates into H + and (OH) - as shown below. When plants decay they release carbon dioxide and the solubility of calcite is favored.Īs noted previously, the "acidity" of a solution is a measure of the amount of H +. Again, a loss of carbon dioxide will result in favoring the precipitation of calcite. Green plants remove carbon dioxide in the process of photosynthesis. This causes to produce more carbonic acid and to shift to the right causing calcite to dissolve. Calcite is not found in sediments in the deep marine environment.Īn increase in load pressure (due to the mass of the overlying material) is to increase the solubility of gases in liquids. In the deep ocean basins (water depths greater than about 11,000 meters), equation is shifted so as to produce more carbonic acid which results in a shift to the right in equation resulting in the solution of calcite. In contrast, carbon dioxide is more soluble in cold water than in warm. Equation will shift to the left to produce more carbonic acid and calcite will precipitate.Ĭalcium carbonate, for example, is often found precipitating in proximity to hot springs. Therefore, if the system is at equilibrium, warming the water will result in a reduction in the amount of the gas, equation will shift so as to produce more CO 2 which results in a decrease in the amount of carbonic acid. 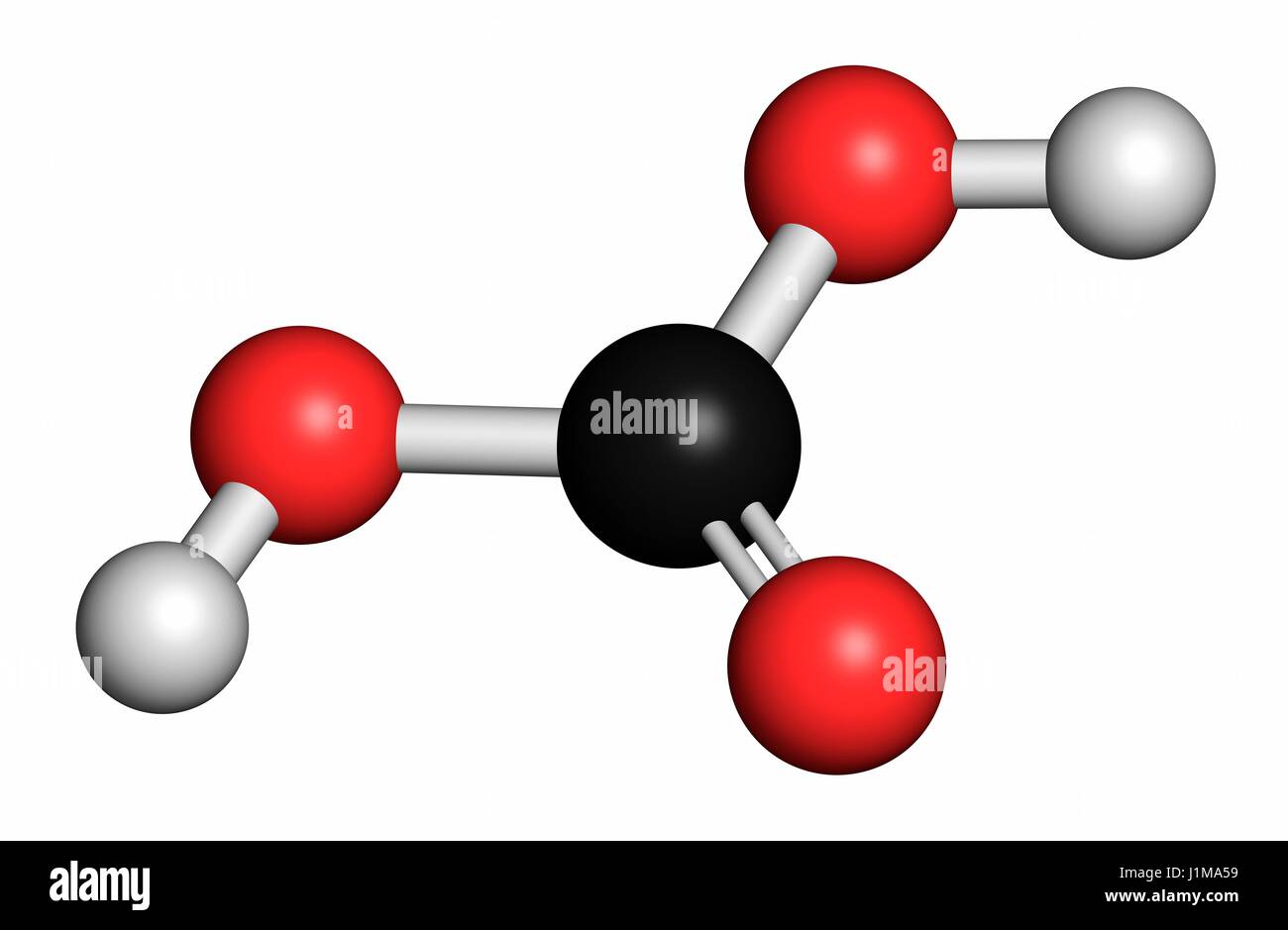
Gases like CO 2 are less soluble in warm solutions than they are in cold solutions. Any process that reduces the amount of CO 2 in the system will cause calcite to precipitate. Therefore, when CO 2 increases calcite goes into solution - dissolves. The increased H 2CO 3 causes reaction to shift to the right which means that CaCO 3 will dissolve.promotes the production of more H 2CO 3 (the equilibrium shifts to use up the increased CO 2).Any process that increases the amount of CO 2: Imagine that this system is at equilibrium. Both equations have their own equilibrium constant. Think of the | | representing equilibrium just as = represents equilibrium. Įquations and clearly relate to each other. The reaction continues until the balance between CO 2 in the air and CO 2 dissolved in water is that given by equation. That is, in reaction the Forward Direction is favored. If we disturb equilibrium by increasing the amount of CO 2 in the air the reaction adjusts by "reversing" the nature of the disturbance - that is, the amount of CO 2 in the air is reduced by dissolving some of the "excess" in water. Is constant at a constant temperature - the so-called equilibrium constant.Ĭhemical equilibrium is dynamic. At a constant temperature the ratio of :ĬO 2 dissolved in water / CO 2 in the air The reaction continues until a balance is established between these two processes and an equilibrium condition is reached. Gaseous CO 2 from the air dissolves in the water and dissolved CO 2 in the water escapes into the air.ĬO 2 in the air = CO 2 dissolved in water Equation, for example could be a system. The system and the surroundings are separated by a "boundary". 
Everything else is part of the surroundings. The Reverse Direction is to the left.Ī System will be defined as some portion of the universe that you are interested. The Forward Direction is to the right as you view this page. 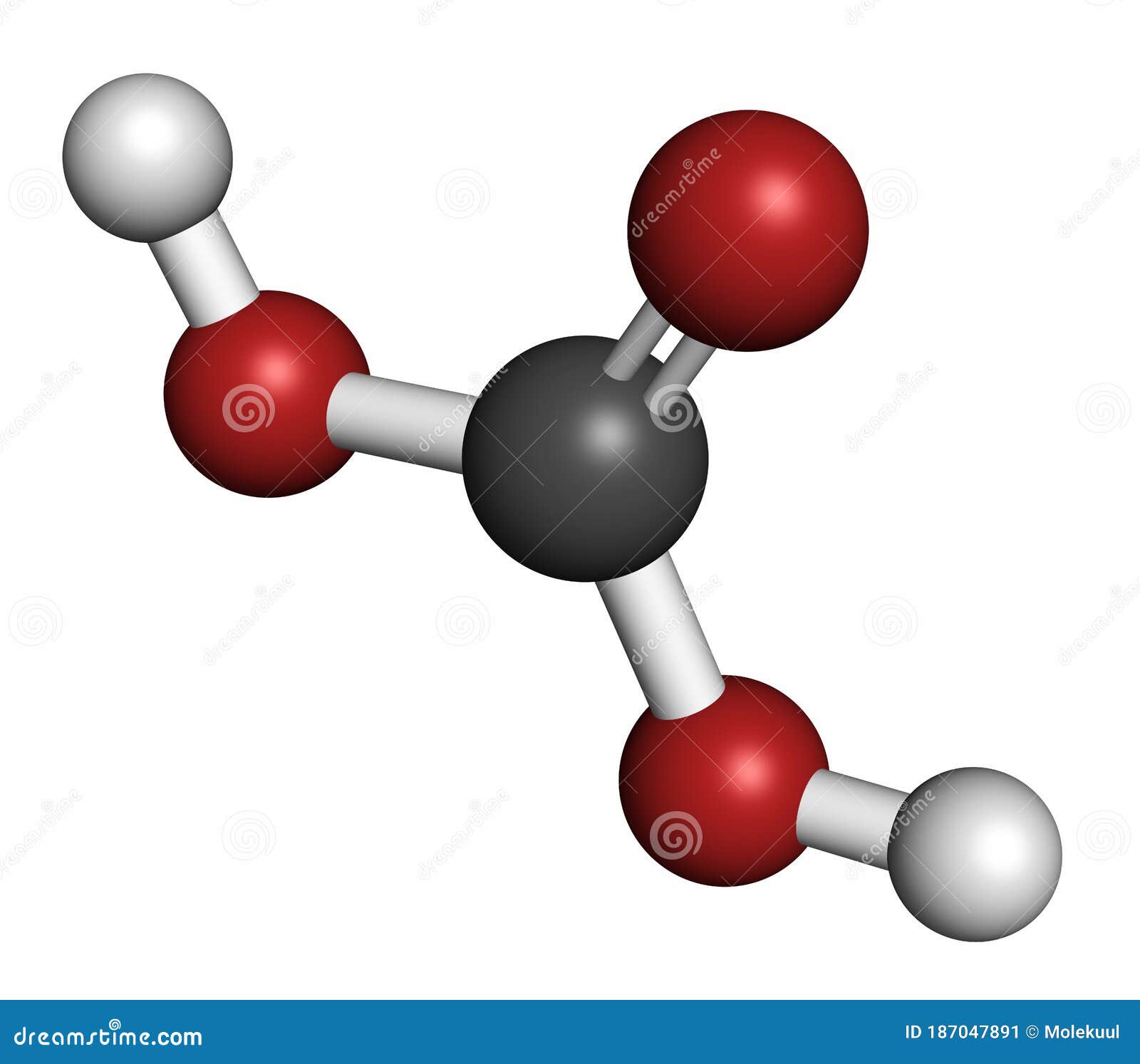
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
